How the Firefly’s Glow Became One of Science’s Greatest Tools
A tiny insect’s magical light called bioluminescence has quietly revolutionised modern medicine, drug discovery, and our understanding of how life works at the cellular level.
That Magical Glow on a Summer Night
You’ve probably seen them tiny sparks of light drifting through the dark, rising from the grass on a warm evening. In South Asia, people call them jugnu. In the West, we call them fireflies or lightning bugs. Children chase them. Poets write about them. And for thousands of years, that mysterious golden glow was simply… magic.
But scientists looked at that same glow and saw something else entirely. They saw a question: How does a living creature produce light without heat, without electricity, and without a flame?
The answer to that question turned out to be one of the most powerful discoveries in modern biology. Today, the firefly’s light or more precisely, the chemistry behind it is used to detect cancer, track infections, test new drugs, and even monitor pollution in rivers and soil. A tiny insect has become one of science’s greatest tools.
Let’s start from the beginning and follow the glow from the firefly’s belly all the way to the hospital lab.

What Is Bioluminescence? (And How Does It Work?)
Bioluminescence simply means “living light.” It’s light produced by a living organism through a chemical reaction happening inside its body. Fireflies do it. So do certain deep-sea fish, glowing mushrooms, and even some bacteria. But fireflies are the most famous and the most useful to scientists.
Here’s the cool part: this light produces almost no heat. Scientists call it “cold light.” Your phone torch gets warm. A candle flame is scorching hot. But the firefly’s glow? You could hold one in your hand and feel nothing because the energy is being converted directly into light, not wasted as heat.
Meet the Key Players
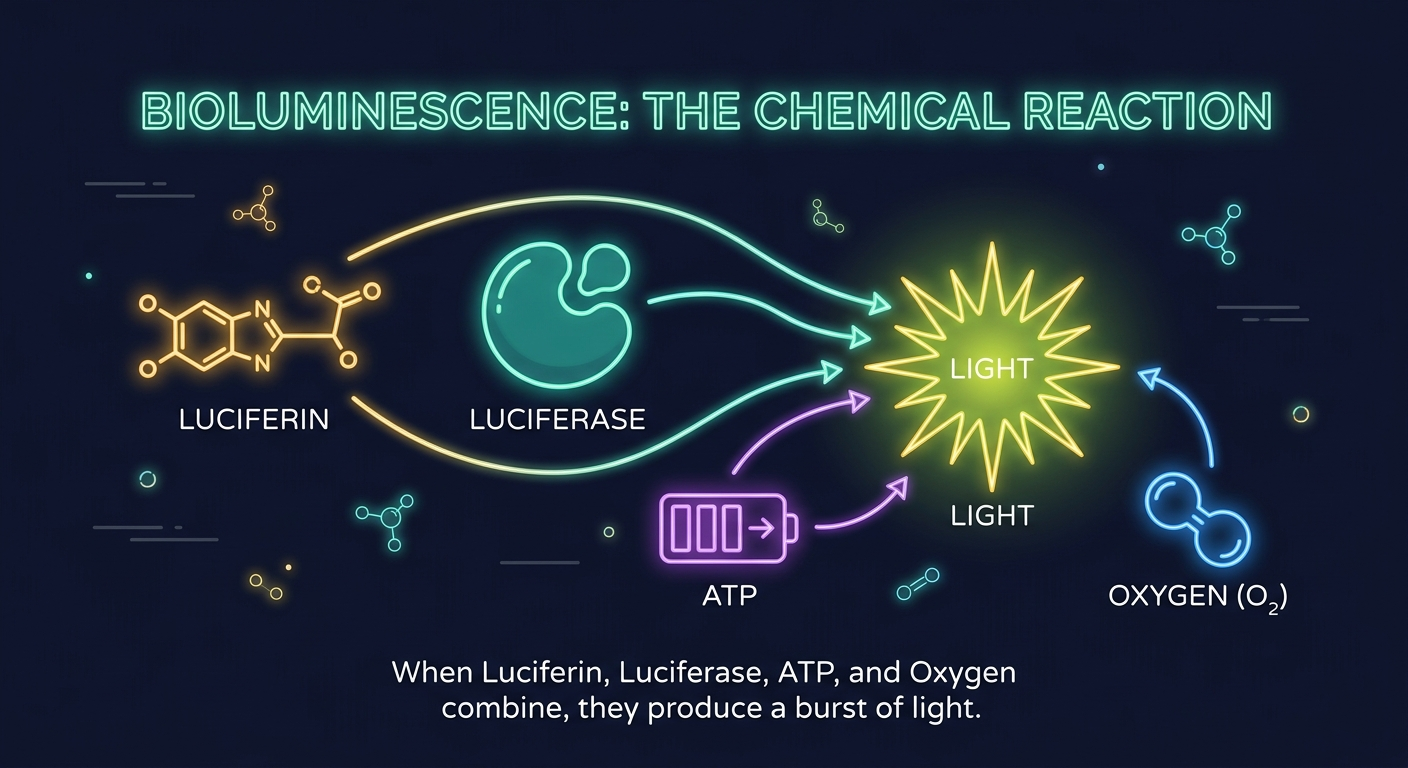
The firefly’s light-making factory uses just four ingredients:
Think of it like baking. Luciferin is the dough the raw material. Luciferase is the oven it transforms the dough into something new. ATP is the electricity powering the oven. And oxygen is the heat that makes the reaction happen. Without any one of these, you don’t get your bread and the firefly doesn’t glow.
Step by Step: How the Glow Happens
Step 1: The firefly’s belly (called the lantern organ) contains special cells packed with the enzyme luciferase.
Step 2: When the firefly wants to flash, it opens tiny air tubes to let oxygen rush in.
Step 3: Luciferase grabs a molecule of luciferin and a molecule of ATP, and combines them with the oxygen.
Step 4: This chemical reaction kicks a tiny particle of light called a photon out of the molecule. That photon is the glow you see.
Step 5: The firefly controls the flash by controlling the oxygen supply like a living light switch!
How Scientists Make Specific Cells Glow
Here’s where things get really exciting. Scientists realised: what if we could take the recipe for making light the luciferase gene and give it to other living cells? What if we could make cancer cells glow? Or brain cells? Or any cell we wanted to watch?
It turns out, we can.
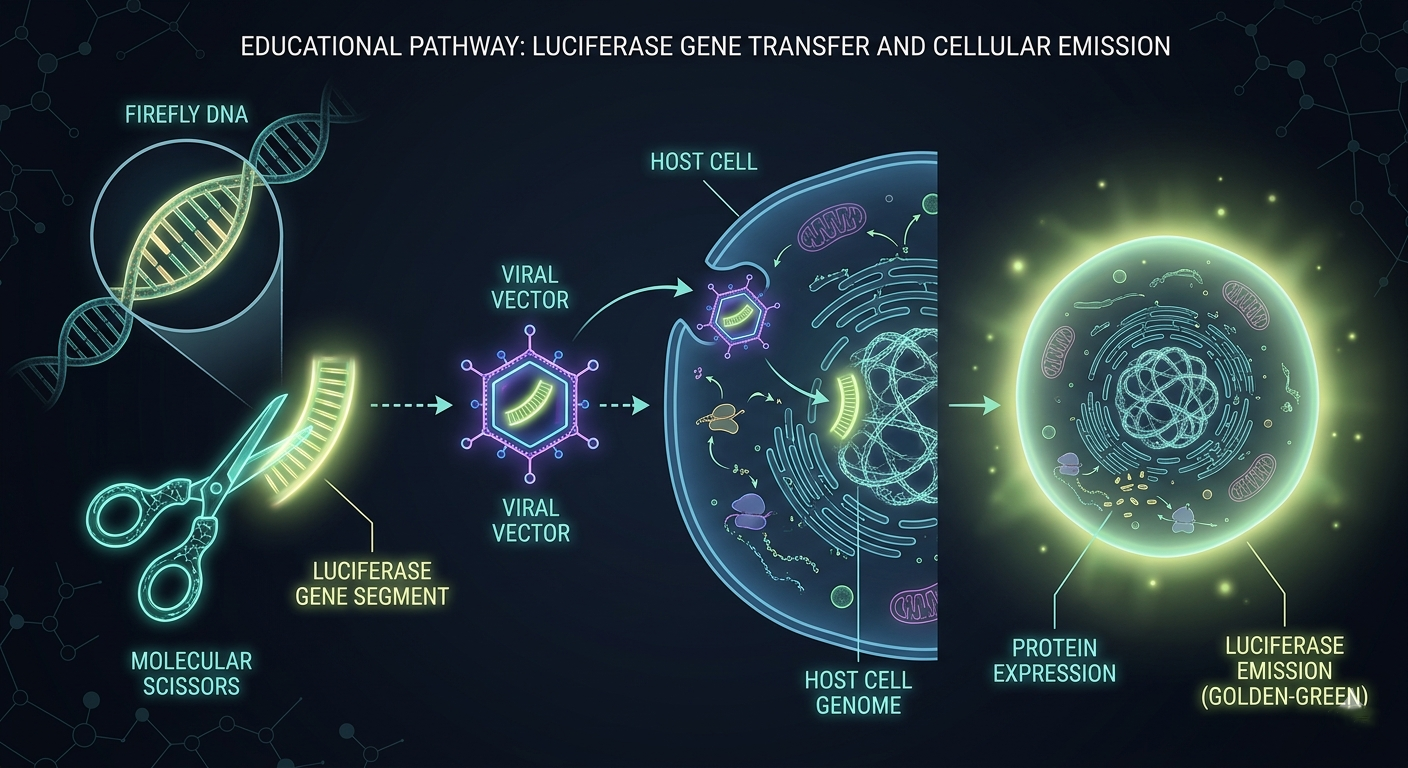
The Luciferase Gene A Biological Blueprint
Every living thing is built using genetic instructions written in DNA. The firefly carries a gene a section of DNA that contains the instructions for making luciferase. Scientists can copy this gene and insert it into any cell they want to study.
This process is called genetic engineering or gene insertion. Think of it like copying a recipe from one cookbook and pasting it into another. The kitchen (the cell) reads the recipe and starts cooking (making luciferase). Once the cell has the luciferase gene, all you need to do is add the fuel (luciferin from outside), and the cell will glow.
The Secret Power of Promoters
Scientists don’t just insert the luciferase gene anywhere. They attach it to a special molecular switch called a promoter.
A promoter is a sequence of DNA that acts like an “ON switch” it only activates the gene it’s attached to under specific conditions. For example, there are promoters that only switch on inside cancer cells, or only inside brain neurons, or only when a certain virus is present.
This means: the luciferase gene only becomes active and the cell only glows when the exact condition the scientist cares about is happening. The light becomes a signal. A message written in photons.
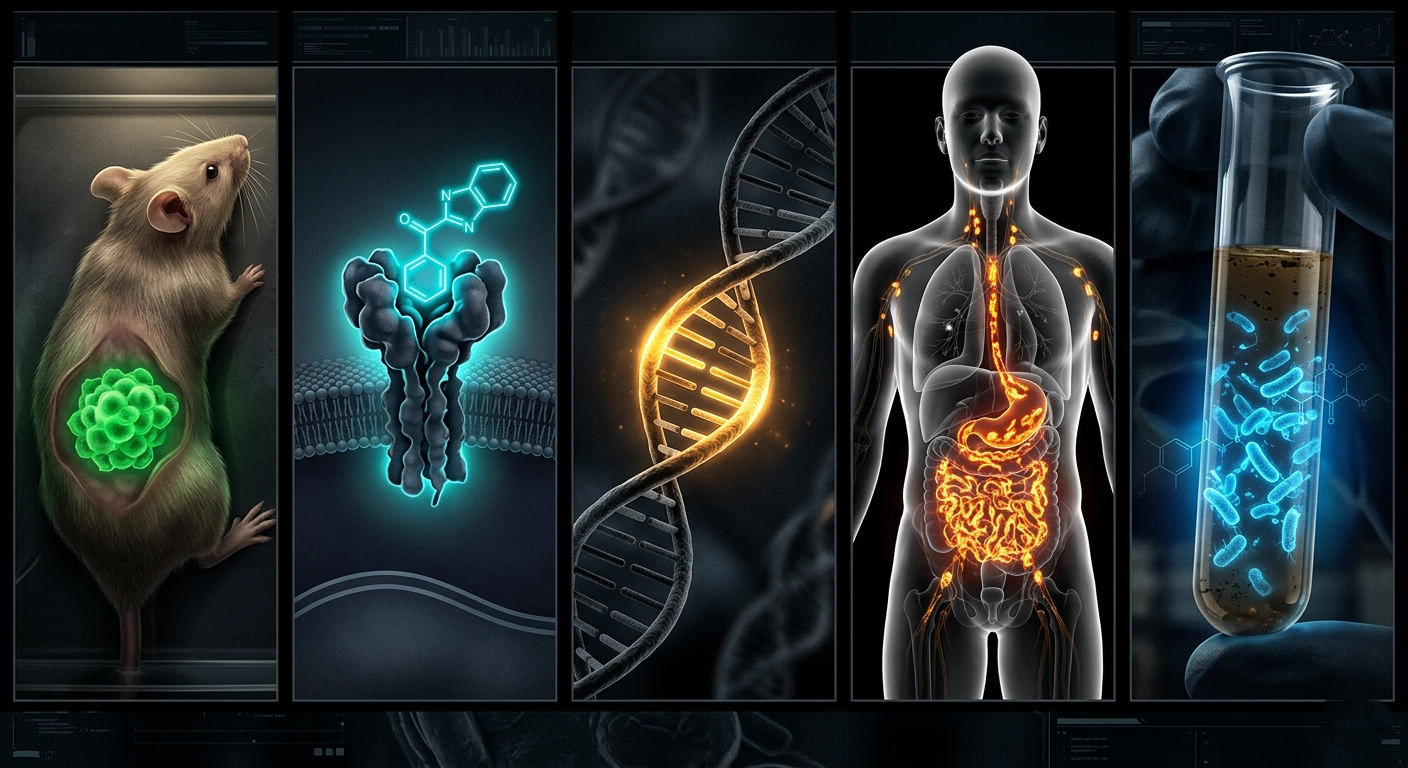
Real-World Examples
Glowing cancer cells: Scientists insert the luciferase gene attached to a cancer-specific promoter into tumour cells. The tumour glows, and researchers can watch it grow, shrink, or spread in a living animal without surgery.
Glowing neurons: Neuroscientists can make nerve cells glow when they fire an electrical signal, essentially watching thoughts happen in real time.
Glowing genes: Scientists attach luciferase to any gene they want to study. When that gene turns on, light turns on. When the gene turns off, light disappears. The light level = gene activity.
Real Research Applications: Where the Glow Goes to Work
The luciferase gene has found its way into laboratories around the world. Here are five powerful ways that firefly light is actively being used in science today.
One particularly powerful application is the luciferase reporter assay a standard lab test used in thousands of pharmaceutical labs worldwide. It uses light output to measure whether a potential drug molecule is activating or blocking a specific gene. It is fast, cheap, and remarkably sensitive.
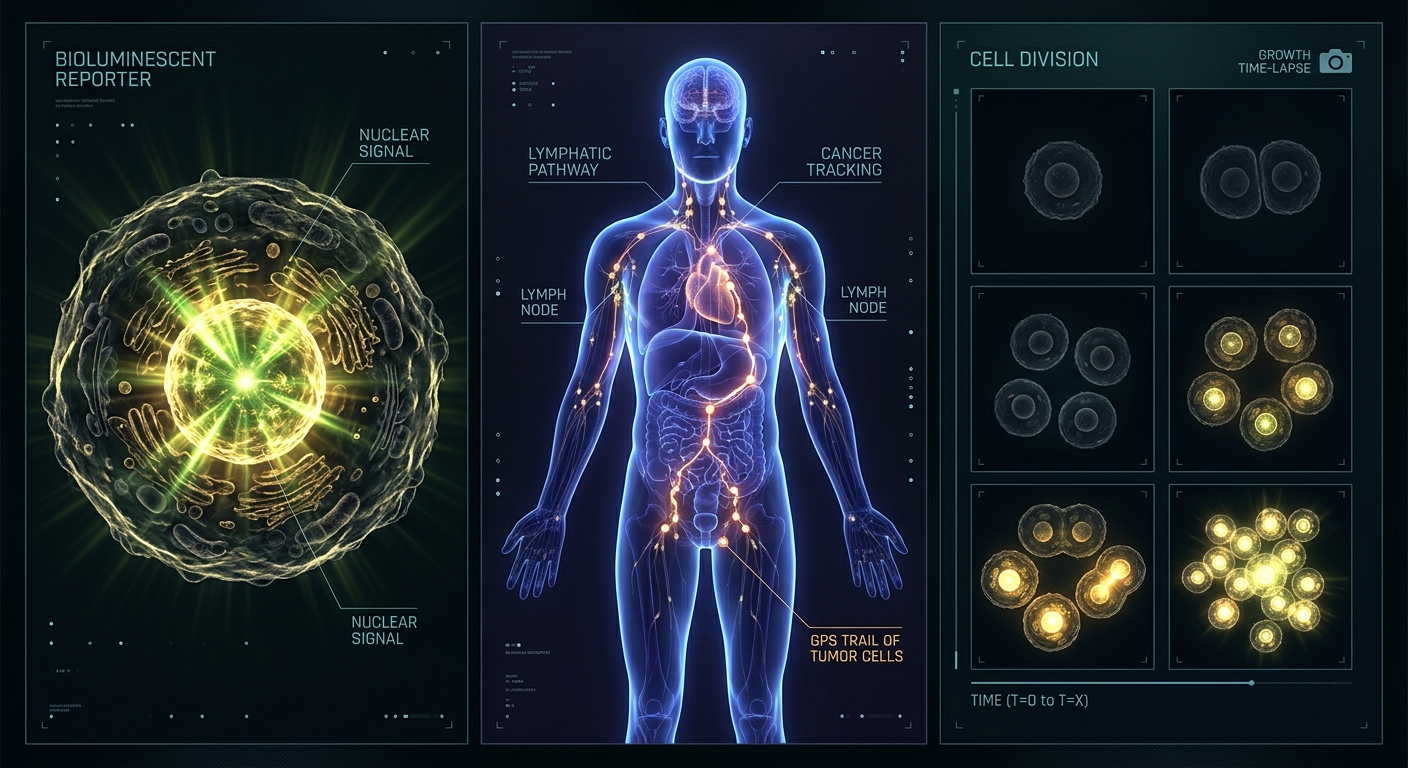
The Microscope, the GPS, and the Camera All in One Glow
Scientists often describe luciferase as the ultimate biological tool because it does the job of three completely different instruments at once.
The Microscope Role: Seeing Without Looking
Traditional microscopes require you to take a sample out of a living organism, stain it with dye, and examine it under glass killing whatever you were studying in the process. Luciferase works differently: the light comes from inside the living cell, in real time. You don’t need to disturb the cell at all. You just wait for it to glow and the amount of light tells you exactly what’s happening molecularly inside it.
The GPS Role: Following Cells Through the Body
In a technique called bioluminescence imaging (BLI), scientists inject glowing tumour cells or bacteria into an animal, then use a highly sensitive camera in a dark chamber to photograph the animal from outside. The glowing regions show exactly where the cells are and by taking photographs over days or weeks, they can follow the journey of every single glowing cell through the whole body, non-invasively.
Think of it like putting a tiny GPS tracker inside every cancer cell. Wherever those cells go lung, liver, brain the signal follows. No surgery required.
The Camera Role: Recording Life Happening
Because luciferase responds to ATP (the cell’s energy molecule), it effectively measures whether a cell is alive and active at any given moment. More glow = more ATP = more cell activity. Scientists studying how drugs affect cells can literally watch the cells respond to treatment in real time, on a screen, like a biological film.
Here’s the deepest insight: light is just detectable. Unlike electrical signals or protein concentrations, light can travel through tissue and be captured by a camera. When you convert a biological event a gene switching on, a cell dividing into light, you’ve translated invisible biology into visible information. That’s what luciferase does: it’s a translator between the molecular world and the human eye.
Why Isn’t It Used Directly in Humans Yet?
At this point you might be wondering: if this is so powerful, why aren’t doctors using glowing cells to diagnose cancer in human patients right now? It’s a fair question and the honest answer is that we’re not quite there yet. Here’s why:
-
Light doesn’t travel well through human tissue. Bioluminescent light is faint, and human tissue (muscle, fat, skin) absorbs and scatters light heavily. In mice which are small and translucent the signal works well. In a human body, the light from deep inside an organ would be nearly impossible to detect at the surface.
-
Getting the luciferase gene into human cells safely is complex. Inserting genes into human cells requires viral vectors or other delivery tools and doing this precisely, without off-target effects, remains a significant challenge in human gene therapy.
-
Luciferin must be delivered to the cells. Unlike in fireflies (which produce their own luciferin), lab cells need luciferin supplied externally. For human use, this would require a safe, reliable delivery method which is still being developed.
-
Ethical and regulatory constraints. Introducing genetic material into living humans requires extensive safety testing, ethical approval, and regulatory oversight. Bioluminescent imaging in humans is still a long-term research goal, not a near-term clinical tool.
-
Sensitivity limits in whole-body imaging. Even in research animals, detecting small numbers of glowing cells (e.g., a tiny cluster of 100 cancer cells) is challenging. Detection technology is improving, but the signal-to-noise problem persists.
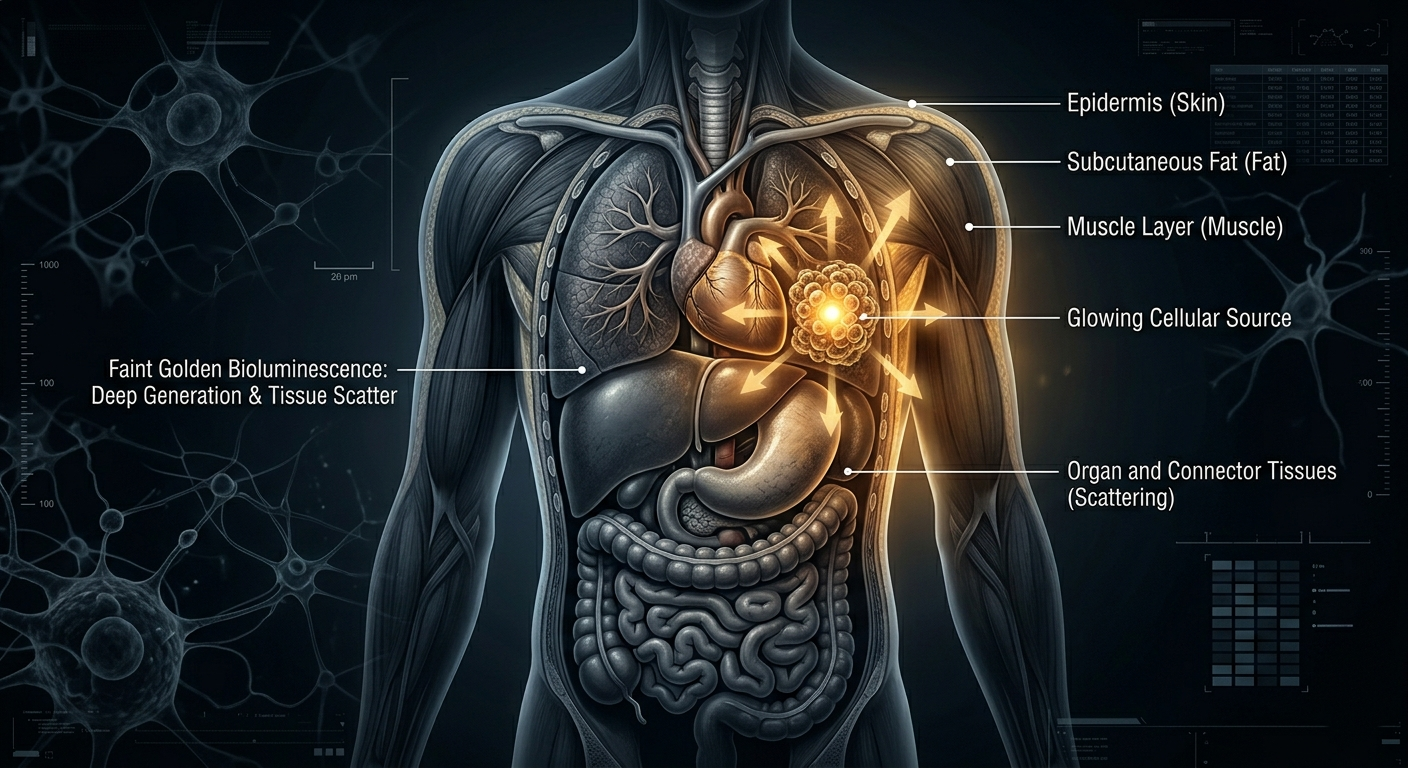
What Comes Next: The Glowing Future
Despite current limitations, the field is advancing rapidly. Researchers around the world are working on exciting new directions that could transform bioluminescence from a laboratory tool into an everyday part of medicine and sustainability.
Glowing Plants as Bio-Lighting
Researchers have already created plants that glow continuously using a modified fungal bioluminescence system. The dream? Streetlamps grown from trees. Sustainable, living light. No electricity required.
Next-Generation Smart Biosensors
Bacteria engineered to glow in response to specific pollutants, pathogens, or chemical signals could become cheap, deployable sensors for environmental monitoring, food safety, and public health surveillance.
Synthetic Bioluminescence Systems
Scientists are engineering entirely new bioluminescent molecules brighter, more stable, with tunable colours that can report on different biological events simultaneously. Different colours for different signals.
Deep-Tissue Medical Imaging
By combining bioluminescence with new near-infrared luciferin variants and more sensitive detectors, researchers hope to one day image bioluminescent signals through human tissue bringing this technology closer to clinical use.
One of the most promising developments is NanoLuc a synthetic luciferase engineered to be 150 times brighter than the original firefly enzyme. Developed by Promega Corporation, NanoLuc is now widely used in drug screening and diagnostics, pushing the limits of what bioluminescent tools can detect.

Nature’s Little Light Now a Big Idea
The story of the firefly and science is a beautiful reminder that nature has already solved many of the problems we’re trying to solve. The firefly didn’t evolve its glow for our benefit it did so to attract mates, warn off predators, and communicate with its own kind. But in doing so, it stumbled upon one of the most elegant chemical reactions in biology.
When a curious scientist asked “how does this work?” and dug into the chemistry of the jugnu’s belly, they unlocked a tool that now helps us fight cancer, develop life-saving drugs, track the spread of viruses, and monitor the health of our planet.
You don’t always need to build something new from scratch. Sometimes, the most powerful technologies are already out there, drifting through the warm summer air, winking at us in the dark.


